Small manufacturing companies face a lot of hurdles these days. Between rising regulatory pressures, such as audits and enforcement letters, and the fact that small manufacturers don’t have the resources that their larger counterparts have access to, owners and plant managers experience added stress and additional duties than what large company owners and managers may face. As a small manufacturer that must comply with U.S. Food and Drug Administration (FDA) regulations, you likely have a tighter budget, limited staff, and diverse product lines. With these factors in mind, you might imagine that generic enterprise software doesn’t do the job for you. You need something tailored more towards your small manufacturing business and its FDA compliance needs.
Take this example: You have a busy, filled shop floor covered with scattered records, manual spreadsheets, and tons of paperwork that can easily get lost and not be followed up on when it comes time for FDA compliance deadlines. You might then have to deal with reactive compliance, i.e., waiting for an audit to happen, as opposed to active compliance, which can be made much easier with the right compliance software.
Compliance software offers so much more than just being a way to check the boxes. This type of monitoring software ensures you keep your compliance deadlines on schedule and you remain in audit trail compliance, for example. The following article highlights FDA regulatory requirements small manufacturers must meet, features of good FDA compliance software, and an implementation guide and cost and ROI considerations of FDA compliance software.

There are many core regulatory requirements that small manufacturing companies must meet, including the following:
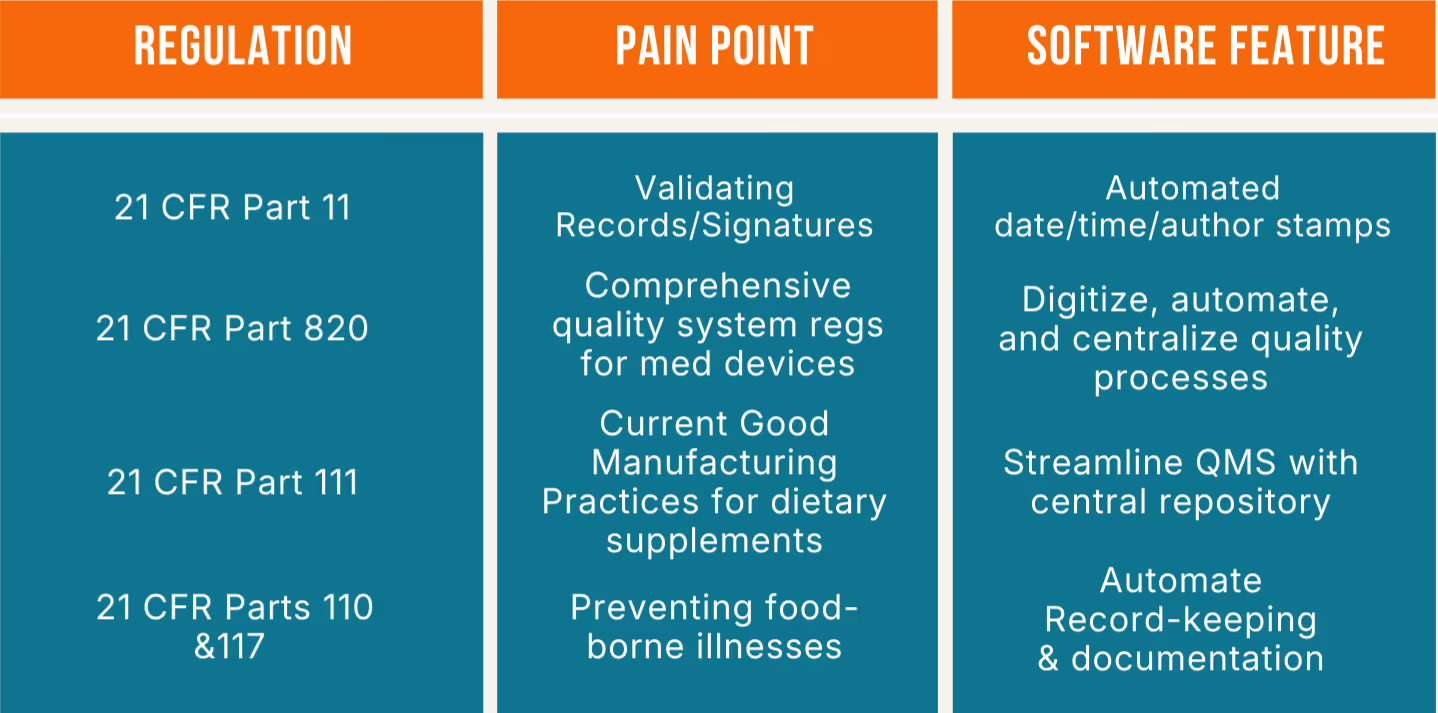
With 21 CFR Part 11, it’s imperative for the company to ensure electronic records/electronic signatures are trustworthy. Compliance software helps by automating these processes every step of the way. To comply with 21 CFR Part 820 (and the ISO 13485 global standard), small businesses must establish and maintain a comprehensive quality management system (QMS), such as corrective and preventive action (CAPA) software and training management processes. Compliance software can help with this task by providing a platform in which to manage the documentation and processes. For 21 CFR part 111 compliance, software automates tasks and streamlines processes along the way. Compliance software helps with the FSMA by ensuring good recordkeeping and documentation and being ready should an audit occur.
For small manufacturers searching for an optimal FDA compliance software program, here are some must-haves and nice-to-haves to help you search:
Consider your industry: When choosing a specific FDA compliance software product, consider your industry and how the software will help you with your industry-specific needs.
Know the benefits of Cloud/SaaS software: Know how cloud-based SaaS may be better suited for your business than a non-cloud offering. For small to medium manufacturers, clou-based software relieves the burden of requiring a server and network/IT expertise to manage the software installation and upgrades.
Consider tech-savvy aspects: Look at the features of the software and consider if they’re better for tech-savvy individuals as opposed to being user-friendly for all.
Don’t over-engineer: Don’t buy more of a compliance software product that you need as doing so could mean greater cost and time factors when having to learn a new system.
If you’re ready to take your current state of spreadsheet chaos to an audit-ready software system, here are some steps:
The benefits of compliance software for a small manufacturing firm are worth it, as long as they require minimal IT assistance and are a win in the cost and time categories.
As a small manufacturer, you should be aware of both the costs and return on investment (ROI) considerations with FDA compliance software. For example, some software solutions have upfront costs including a software fees, implementation/configuration costs, and money expended for training, ongoing support, and data migration. Look for products like MakerComply, which offers a simple pricing structure and no implementation fees.
On the plus side, look at ROI benefits, such as spending less time on audits, fewer fines and warning letters due to regulatory noncompliance, reduced scrap and recall risk, improved operational visibility, and supplier compliance. If you sell products to other manufacturers, you know there's a clear link between FDA compliance and customer retention
When you compare the benefits with the costs, you’ll find that it pays to be in compliance with help from FDA compliance software, especially if you choose a cost-effective solution.
When the time comes to choose your compliance software vendor, consider the following questions:
Considering and asking these questions will help you choose the right vendor and also ensure that your compliance software will continue to be the best option in the future.

1. What exactly qualifies as “FDA compliance software” for a small manufacturer?
An electronic quality management system (eQMS) that automates, manages, and documents processes per FDA regulations. You can use individual software modules that get you closer to that goal, for example, MakerComply offers modules for Free Training Tracking, full Training Management, Incident Management with CAPA, Document Management, and Audit Management.
2. Does my manufacturing business have to use software, or can we still rely on spreadsheets and paper?
You can still use spreadsheets and paper but there will be an added burden on your business in order to comply with strict FDA requirements and regulations pertaining to record keeping.
3. Which FDA regulations should my small manufacturing firm worry about?
Small manufacturing companies must comply with all FDA regulations pertinent to their business and area of industry.
4. How long does it typically take a small manufacturer to implement compliance software?
The length of time it takes for a small manufacturer to implement compliance software varies and depends on the vendor offerings. In general, this type of implementation may take anywhere from a month to a year.
5. How much does an FDA-compliance software solution cost for a small manufacturing company?
FDA compliance software costs vary from vendor to vendor. However, while larger manufacturing companies could pay up to $100,000 for a custom software solution., smaller companies can take advantage of tools like MakerComply, at a fraction of that price.
6. What are the risks of choosing the wrong software (or no software) for compliance?
FDA regulations may impose fines or warning letters for lack of compliance software, depending on the requirement. In addition, the business may face other financial, legal, and reputation-related risks.
7. How can I measure ROI or the business benefit of compliance software?
To measure the ROI of compliance software, you can use the following formula: ROI= (Net Benefits - Costs) / Costs x 100%. Net benefits can include reduced fines, labor savings, improved reputation, and future business opportunities as financial gains. Costs may include the software price and costs of implementation, training, and maintenance.
8. What should I check for before purchasing a compliance software package?
Ensure that the compliance software package can be integrated into your current processes and will be affordable and practical for your factory.
11. Are cloud-based solutions safe and acceptable for FDA compliance (vs on-premises)?
All FDA requirements for security, validation, and data integrity must be met for cloud-based solutions to be safe and acceptable.
References:
Small manufacturing companies face a lot of hurdles these days. Between rising regulatory pressures, such as audits and enforcement letters, and the fact that small manufacturers don’t have the resources that their larger counterparts have access to, owners and plant managers experience added stress and additional duties than what large company owners and managers may face. As a small manufacturer that must comply with U.S. Food and Drug Administration (FDA) regulations, you likely have a tighter budget, limited staff, and diverse product lines. With these factors in mind, you might imagine that generic enterprise software doesn’t do the job for you. You need something tailored more towards your small manufacturing business and its FDA compliance needs.
Take this example: You have a busy, filled shop floor covered with scattered records, manual spreadsheets, and tons of paperwork that can easily get lost and not be followed up on when it comes time for FDA compliance deadlines. You might then have to deal with reactive compliance, i.e., waiting for an audit to happen, as opposed to active compliance, which can be made much easier with the right compliance software.
Compliance software offers so much more than just being a way to check the boxes. This type of monitoring software ensures you keep your compliance deadlines on schedule and you remain in audit trail compliance, for example. The following article highlights FDA regulatory requirements small manufacturers must meet, features of good FDA compliance software, and an implementation guide and cost and ROI considerations of FDA compliance software.

There are many core regulatory requirements that small manufacturing companies must meet, including the following:

With 21 CFR Part 11, it’s imperative for the company to ensure electronic records/electronic signatures are trustworthy. Compliance software helps by automating these processes every step of the way. To comply with 21 CFR Part 820 (and the ISO 13485 global standard), small businesses must establish and maintain a comprehensive quality management system (QMS), such as corrective and preventive action (CAPA) software and training management processes. Compliance software can help with this task by providing a platform in which to manage the documentation and processes. For 21 CFR part 111 compliance, software automates tasks and streamlines processes along the way. Compliance software helps with the FSMA by ensuring good recordkeeping and documentation and being ready should an audit occur.
For small manufacturers searching for an optimal FDA compliance software program, here are some must-haves and nice-to-haves to help you search:
Consider your industry: When choosing a specific FDA compliance software product, consider your industry and how the software will help you with your industry-specific needs.
Know the benefits of Cloud/SaaS software: Know how cloud-based SaaS may be better suited for your business than a non-cloud offering. For small to medium manufacturers, clou-based software relieves the burden of requiring a server and network/IT expertise to manage the software installation and upgrades.
Consider tech-savvy aspects: Look at the features of the software and consider if they’re better for tech-savvy individuals as opposed to being user-friendly for all.
Don’t over-engineer: Don’t buy more of a compliance software product that you need as doing so could mean greater cost and time factors when having to learn a new system.
If you’re ready to take your current state of spreadsheet chaos to an audit-ready software system, here are some steps:
The benefits of compliance software for a small manufacturing firm are worth it, as long as they require minimal IT assistance and are a win in the cost and time categories.
As a small manufacturer, you should be aware of both the costs and return on investment (ROI) considerations with FDA compliance software. For example, some software solutions have upfront costs including a software fees, implementation/configuration costs, and money expended for training, ongoing support, and data migration. Look for products like MakerComply, which offers a simple pricing structure and no implementation fees.
On the plus side, look at ROI benefits, such as spending less time on audits, fewer fines and warning letters due to regulatory noncompliance, reduced scrap and recall risk, improved operational visibility, and supplier compliance. If you sell products to other manufacturers, you know there's a clear link between FDA compliance and customer retention
When you compare the benefits with the costs, you’ll find that it pays to be in compliance with help from FDA compliance software, especially if you choose a cost-effective solution.
When the time comes to choose your compliance software vendor, consider the following questions:
Considering and asking these questions will help you choose the right vendor and also ensure that your compliance software will continue to be the best option in the future.
1. What exactly qualifies as “FDA compliance software” for a small manufacturer?
An electronic quality management system (eQMS) that automates, manages, and documents processes per FDA regulations. You can use individual software modules that get you closer to that goal, for example, MakerComply offers modules for Free Training Tracking, full Training Management, Incident Management with CAPA, Document Management, and Audit Management.
2. Does my manufacturing business have to use software, or can we still rely on spreadsheets and paper?
You can still use spreadsheets and paper but there will be an added burden on your business in order to comply with strict FDA requirements and regulations pertaining to record keeping.
3. Which FDA regulations should my small manufacturing firm worry about?
Small manufacturing companies must comply with all FDA regulations pertinent to their business and area of industry.
4. How long does it typically take a small manufacturer to implement compliance software?
The length of time it takes for a small manufacturer to implement compliance software varies and depends on the vendor offerings. In general, this type of implementation may take anywhere from a month to a year.
5. How much does an FDA-compliance software solution cost for a small manufacturing company?
FDA compliance software costs vary from vendor to vendor. However, while larger manufacturing companies could pay up to $100,000 for a custom software solution., smaller companies can take advantage of tools like MakerComply, at a fraction of that price.
6. What are the risks of choosing the wrong software (or no software) for compliance?
FDA regulations may impose fines or warning letters for lack of compliance software, depending on the requirement. In addition, the business may face other financial, legal, and reputation-related risks.
7. How can I measure ROI or the business benefit of compliance software?
To measure the ROI of compliance software, you can use the following formula: ROI= (Net Benefits - Costs) / Costs x 100%. Net benefits can include reduced fines, labor savings, improved reputation, and future business opportunities as financial gains. Costs may include the software price and costs of implementation, training, and maintenance.
8. What should I check for before purchasing a compliance software package?
Ensure that the compliance software package can be integrated into your current processes and will be affordable and practical for your factory.
11. Are cloud-based solutions safe and acceptable for FDA compliance (vs on-premises)?
All FDA requirements for security, validation, and data integrity must be met for cloud-based solutions to be safe and acceptable.
References:
